flutolanil (bsi,draft e-iso,draft f-iso)
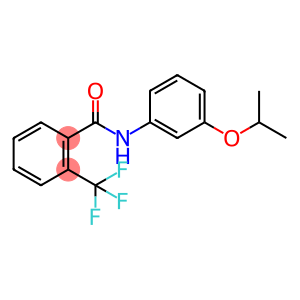
FLUTOLANIL
CAS: 66332-96-5
Molecular Formula: C17H16F3NO2
flutolanil (bsi,draft e-iso,draft f-iso) - Names and Identifiers
| Name | FLUTOLANIL |
| Synonyms | MONCUT NNF 136 MONCUT(R) FLUTOLANIL Flutalanil flutolanil (bsi,draft e-iso,draft f-iso) 3'-Isopropoxy-2-trifluoromethylbenzanilide N-[3-(Isopropyloxy)phenyl]-2-(trifluoromethyl)benzamide |
| CAS | 66332-96-5 |
flutolanil (bsi,draft e-iso,draft f-iso) - Physico-chemical Properties
| Molecular Formula | C17H16F3NO2 |
| Molar Mass | 323.31 |
| Density | 1.2463 (estimate) |
| Melting Point | 108° (Araki, Yabutani); mp 104-105° (Araki, 1985) |
| Boling Point | 339.1±42.0 °C(Predicted) |
| Water Solubility | 6.53 mg l-1 (20 °C) |
| Solubility | Chloroform (Slightly), Methanol (Slightly) |
| Vapor Presure | 6.5 x 10-6 Pa (25 °C) |
| Appearance | Solid |
| Color | White to Pale Orange |
| pKa | 12.44±0.70(Predicted) |
| Storage Condition | 0-6°C |
| Physical and Chemical Properties | Pure product is white odorless crystal. M. p.108 ℃(102~103 ℃), vapor pressure 1.77 × 10-9Pa (20 ℃). The solubility at 20 ℃ is: acetone 642g/L, methanol 606g/L (480g/L), chloroform 341g/L (238g/L), toluene 56g/L (65g/L), xylene 29g/L, hexane 3g/L, water 9.6mg/L. The partition coefficient is 3.7, stable in aqueous solution with pH value of 3~9, no decomposition when heated at 100 ℃ for 5h or 50 ℃ for 14d, and the decomposition rate is 1% when irradiated by fluorescent lamp (17000lx, 96h), indicating good stability to heat and light. The half-life in soil is 40~60 days. |
flutolanil (bsi,draft e-iso,draft f-iso) - Risk and Safety
| RTECS | CV5581320 |
| Toxicity | LD50 in male and female rats, male and female mice (mg/kg): >10,000, >10,000 orally; in male and female rats (mg/kg): >5000 dermally (Araki) |
flutolanil (bsi,draft e-iso,draft f-iso) - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| toxicity | Acute oral LD50>10000mg/kg in rats and mice, acute skin at a dose of 5000mg/kg showed no toxicity, the skin and mucous membrane irritation is very slight. Carp lc502.4 mg/L(48h). |
| Use | belongs to benzamide derivatives, with preventive and therapeutic effects of systemic fungicide. For the control of certain fungi of Basidiomycetes. It has a strong inhibitory effect on the growth of the mycelium and the formation of the invasion of the mycelium. Such as control of Rhizoctonia caused by rice sheath blight, with 50mg/L concentration of stems and leaves treatment, control effect of 100%. The systemic fungicide is effective for rice sheath blight and potato nevus. |
| production method | O-trifluoromethyl chlorobenzene is obtained by Grignard reaction, Carbonylation and hydrolysis to O-trifluoromethyl benzoic acid, then the acylation reaction was carried out, and then the reaction with M-isopropoxyaniline was carried out to prepare fluorine supported bacteria. preparation method-o-trifluoromethyl benzoyl chloride O-trifluoromethyl chlorobenzene can be used as raw material, after Grignard reaction, then carbonylation reaction with dry ice, and then hydrolysis to get O-trifluoromethyl benzoic acid, the acid is acylated to give O-trifluoromethyl benzoyl chloride. O-trifluoromethyl benzoyl chloride was added to tetrahydrofuran under ice-cooling, and reacted with M-isopropoxyaniline in the presence of triethylamine at room temperature for 2H to obtain fluorophanin. Preparation Method difluoromethylbenzamido n can also be obtained by reacting 3-(2-trifluoromethylbenzoylamino) phenol with 2-chloropropane. |
| category | pesticide |
| toxicity grade | low toxicity |
| Acute toxicity | oral-rat LD50: 10000 mg/kg; Oral-mouse LD50: 10000 mg/kg |
| flammability hazard characteristics | flammable; Toxic nitrogen oxides and chloride gases from combustion |
| storage and transportation characteristics | The warehouse is ventilated and dried at low temperature; It is stored and transported separately from food raw materials |
| extinguishing agent | dry powder, foam, sand |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: FLUTOLANIL Request for quotation
CAS: 66332-96-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 66332-96-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Flutolanil Visit Supplier Webpage Request for quotationCAS: 66332-96-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: FLUTOLANIL Visit Supplier Webpage Request for quotationCAS: 66332-96-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: FLUTOLANIL Request for quotation
CAS: 66332-96-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 66332-96-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Flutolanil Visit Supplier Webpage Request for quotationCAS: 66332-96-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: FLUTOLANIL Visit Supplier Webpage Request for quotationCAS: 66332-96-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


